+1 800-123-1234
Only used in cases of accidents, illnesses, etc. that are not life-threatening
For more serious problems please call 911.
511 SW 10th Ave 1206, Portland, OR United States
+1 800-123-1234
Only used in cases of accidents, illnesses, etc. that are not life-threatening
For more serious problems please call 911.
511 SW 10th Ave 1206, Portland, OR United States
+1 800-123-1234
Only used in cases of accidents, illnesses, etc. that are not life-threatening
For more serious problems please call 911.
511 SW 10th Ave 1206, Portland, OR United States


Specialized Consumables for Assisted Reproduction (Medical Device)
Registration Certificate No.: GDMDR 20232181838
Model: Center-well Culture Dish, 35/60/90mm Culture Dish (Flat Bottom), Four-well Culture Plate
Product Information
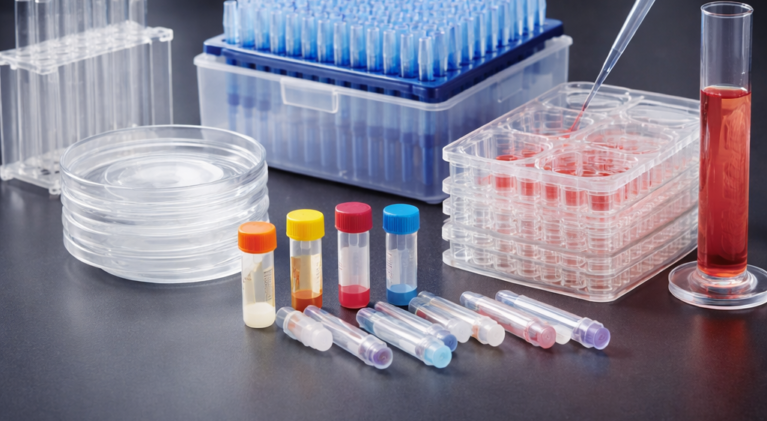
In vitro fertilization (IVF) refers to the process of taking sperm and eggs out of the body and completing the fertilization process in an artificially controlled environment. Jet Biofil’s Specialized Consumables for Assisted Reproduction are specifically designed to provide safe and reliable products for complex IVF and other assisted reproductive applications. Through rigorous testing, including authoritative third-party biocompatibility inspection, in vitro mouse embryo experiments, and human sperm survival tests, ensuring the vitality of human reproductive cells and embryos throughout the complex processes of preparation, storage, operation, cultivation, and transfer in an in vitro environment
Center-well culture dish
Flat bottom 35/60/90 mm culture dish
Four-well culture plate
PES Ultrafiltration Membranes Features
Features
Features
Preferred Raw Materials
Medical-grade raw materials that conform to USP Class VI standards are preferred, with well-defined composition and stable performance, and free from any animal-derived components.
Reliable Quality
Stringent production and quality control is implemented as per ISO13485 and GMP standards to ensure product quality
High Cleanliness
Production takes place within a controlled environment meeting class 100,000 cleanroom standards, ensuring a scientifically rigorous and sterile manufacturing setting
Competitive Stability
We have achieved highly automated production to maximize the separation between human and machine, thus minimizing errors caused by manual interventions
Safety Ensured
The product undergoes validation by an accredited third-party testing institute to ensure compliance with relevant standards for extractables, biocompatibility, and biological safety
Traceability
Each product is individually packaged in clean medical-grade outer packaging that is easy to tear open. Every item is labeled with a lot number to ensure precise traceability of quality
Centrifuge Tubes
| Cat. No | Capacity (mL) | Bottom Type | Maximum RCF (×g) | Packaging | Sterile | Qty Per Bag | Qty Per Case |
|---|---|---|---|---|---|---|---|
| CSP020015 | 15 | Conical | 12,000 | Triple-bagged packaging | Y | 25 | 500 |
| CSP020050 | 50 | Conical | 12,000 | Triple-bagged packaging | Y | 25 | 500 |
Serological Pipets
| Cat. No | Capacity (mL) | Scale (mL) | Colour Code | Packaging | Sterile | Qty Per Bag | Qty Per Case |
|---|---|---|---|---|---|---|---|
| CSP010005 | 5 | 1/10 | Blue | Triple-bagged packaging | Y | 10 | 200 |
| CSP010010 | 10 | 1/10 | Orange | Triple-bagged packaging | Y | 10 | 200 |
| CSP010025 | 25 | 1/10 | Red | Triple-bagged packaging | Y | 10 | 150 |
| CSP010050 | 50 | 1/10 | Purple | Triple-bagged packaging | Y | 10 | 100 |
IVF-Specialized Centre-well Culture Dish And Culture Dish
| Cat. No | Model | Description | Surface Type | Sterile | Qty Per Bag | Qty Per Case |
|---|---|---|---|---|---|---|
| IVF050060 | Centre-well | IVF-Specialized Center-well Culture Dish | Non-treated | Y | 10 | 600 |
| IVF050035 | 35 mm, flat bottom | IVF-Specialized 35 mm Culture Dish | Non-treated | Y | 10 | 960 |
| IVF051060 | 60 mm, flat bottom | IVF-Specialized 60 mm Culture Dish | Non-treated | Y | 10 | 600 |
| IVF050090 | 90 mm, flat bottom | IVF-Specialized 90 mm Culture Dish | Non-treated | Y | 10 | 500 |
| IVF041004 | Four-well plate | IVF-Specialized Four-well Culture Plate | Non-treated | Y | 1 | 100 |
Frequently Asked Questions
Size information of commonly used microwell plates?
What is the chemical resistance of microwell plate?
What is the range of centrifugal force that the microwell plate can withstand?
Does the deep well plate can be applied to cell culture?
 English
English
 KR
KR  IA
IA  JP
JP