

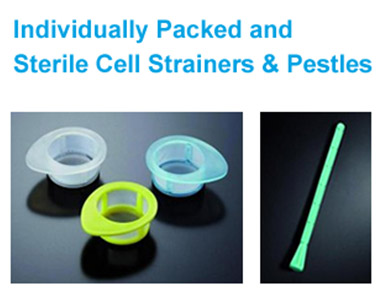
Cell strainers are manufactured from a strong nylon mesh with evenly spaced mesh pores and are gamma resistant.
These cell strainers are sterile, rapid, easy-to-use devices for isolating primary cells to consistently obtain a uniform single-cell suspension from tissues.
Protect your valuable flow cytometry and cell sorting instrumentation by reliably removing clumps and debris from cell suspensions and clinical samples prior to analysis.
 Available in 3 mesh sizes: 40 µm,70 µm and 100 µm
Available in 3 mesh sizes: 40 µm,70 µm and 100 µm  3 different colors: blue, white, and yellow, for easy identification.
3 different colors: blue, white, and yellow, for easy identification.  Improved uniformity of single cell suspensions
Improved uniformity of single cell suspensions  Made of a strong nylon mesh with evenly spaced mesh pores
Made of a strong nylon mesh with evenly spaced mesh pores  The extended lip on the strainer enables aseptic handling with forceps
The extended lip on the strainer enables aseptic handling with forceps  Designed to fit perfectly into a JET 50 ml conical tube
Designed to fit perfectly into a JET 50 ml conical tube  Ready-to-use, sterilized by gamma irradiation
Ready-to-use, sterilized by gamma irradiation  Individually packaged
Individually packaged  DNase-& RNase-free
DNase-& RNase-free  Non-pyrogenic
Non-pyrogenic

 Make using PP Materials with high hardness and wear resistance
Make using PP Materials with high hardness and wear resistance  Comes with non-slip and easy-grip handle for better griding and reduced sample loss
Comes with non-slip and easy-grip handle for better griding and reduced sample loss Non-Pyrogenic and certified DNase-& RNase-free
Non-Pyrogenic and certified DNase-& RNase-free
| Cell Strainer 40 μm | Cell Strainer 70 μm | Cell Strainer 100 μm | |
|---|---|---|---|
| Material | Nylon mesh | Nylon mesh | Nylon mesh |
| Extended lip on Strainer | Yes | Yes | Yes |
| Packing | ind. packed | ind. packed | ind. packed |
| Purity grade(s) | Sterile, pyrogen-, DNase-, RNase-, human and bacterial DNA-free. Non-cytotoxic | Sterile, pyrogen-, DNase-, RNase-, human and bacterial DNA-free. Non-cytotoxic | Sterile, pyrogen-, DNase-, RNase-, human and bacterial DNA-free. Non-cytotoxic |
| Sterilization | 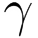 - irradiation - irradiation |
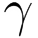 - irradiation - irradiation |
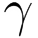 - irradiation - irradiation |
| Cat. No. | Pore size(μm) | Color | Sterile | Qty.per bag/case |
|---|---|---|---|---|
| CSS010040 | 40 | Blue | Y | 1/50 |
| CSS010070 | 70 | Natural | Y | 1/50 |
| CSS010100 | 100 | Yellow | Y | 1/50 |
| CSP001001 (Pestles) | 13.5cm | Fluorescent green | Y | 1/100 |
Cell Strainers are manufactured in accordance with ISO13485:2003 & ISO9001:2008 quality management systems in class 100,000 clean room. Radiation protocols, Quality certificate and Certificate of Compliance are provided with every shipment upon request

| Component Material | Nylon mesh |
| Sterilization Assurance Level | SAL 10 ‾6. Every lot has been irradiated and dosimetrically released. Procedures for determining the dosage comply with ISO11137 |
| Non-Pyrogenic | Products are validated to have less than 0.5EU/ml |
| Cytotoxicity | All material resins are qualified using USP and /or ISO 10993 standards for cytotoxicity and have been shown to be non-toxic |
| DNase & RNase Free | Products are labeled DNase & RNase free and have been validated |
| Quality Control Testing | Representative samples have been selected from each lot and inspected according to ZK-QI |
Cell Strainers are individually packed in easy-to-open bag to assure freedom from contamination. Every case is printed with lot No. for quality traceability as well as expiry date. We warrant that the packaging is sufficient to protect the products against any damage during transit and storage.

Cell and Tissue culture products series are all innovatively designed by JET Engineers and manufactured under the control of ISO 9001: 2008 and ISO 13485 quality management systems. All the JET Biofil product are manufactured with 100% USP VI crystal class virgin polystyrene ( GPPS) and equal high quality polyethylene ( PE) to eliminate all extraneous materials and ensure the integrity. Furthermore, the high transparency of high class material ensures good observability. In addition, all the products are sterilized by gamma irradiation, certified DNase RNase free and non- Pyrogenic. Products are modified with vacuum gas plasma, causing the very hydrophobic polystyrene surface to become negatively charged and hydrophilic, allowing the cells to attach evenly and consistently.

In addition to this, BioStar Lifetech Offers a Complete Satisfaction Guarantee So You Can Be Confident of Your Purchasing Decision. If, For Any Reason You Are Not Satisfied With The Product Performance Or Service Provided, We Will Either Replace Or Issue A Refund For The Purchase Price Of Your Product.
